Further Information
Alternate Names/Synonyms:
Angiopoietin-like Protein 4; FIAF; Fasting-induced Adipose Factor; HFARP; Hepatic Fibrinogen/Angiopoietin-related Protein
See on label.
32160000
liquid
Liquid. 0.2µm-filtered solution in PBS, pH 7.4. Contains no preservatives.
After opening, prepare aliquots and store at -20°C.Avoid freeze/thaw cycles.
Recombinant human coiled-coil domain of ANGPTL4.
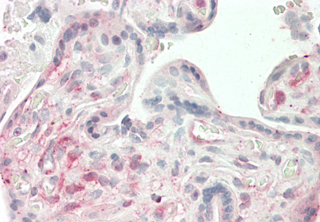
Monoclonal Antibody. Recognizes the coiled-coil domain (CCD) of human ANGPTL4. Detects a band of ~19kDa by Western blot. Isotype: Mouse IgG1kappa. Clone: Kairos4-397G. Applications: ELISA, IHC, WB. Liquid. 0.2µm-filtered solution in PBS, pH 7.4. Contains no preservatives. ANGPTL4 mainly expressed in endothelial cells (hypoxia-induced). Regulates angiogenesis and modulates tumorgenesis and directly regulates lipid, glucose, and energy metabolism. Inhibits proliferation, migration, and tubule formation of endothelial cells and reduces vascular leakage. ANGPTL4 is a protein consisting of an N-terminal coiled-coil domain and a C-terminal fibrinogen-like domain (FLD). Both domains have distinct biological functions. The coiled-coil domain is responsible for the inhibitory effects on lipoprotein lipase (LPL) converting the active form of LPL into an inactive form, and the FLD domain mediates its antiangiogenic functions. The coiled coil and the FLD domains are separated by a short linker that can be cleaved after secretion. ANGPTL4 appears on the cell surface as the full-length form, where it can be released by heparin treatment. ANGPTL4 protein is then proteolytically cleaved by proprotein convertases (PCs), including furin, PC5/6, paired basic amino acid-cleaving enzyme 4, and PC7.
Q9BY76
Plastic Vial
ANGPTL4 mainly expressed in endothelial cells (hypoxia-induced). Regulates angiogenesis and modulates tumorgenesis and directly regulates lipid, glucose, and energy metabolism. Inhibits proliferation, migration, and tubule formation of endothelial cells and reduces vascular leakage. ANGPTL4 is a protein consisting of an N-terminal coiled-coil domain and a C-terminal fibrinogen-like domain (FLD). Both domains have distinct biological functions. The coiled-coil domain is responsible for the inhibitory effects on lipoprotein lipase (LPL) converting the active form of LPL into an inactive form, and the FLD domain mediates its antiangiogenic functions. The coiled coil and the FLD domains are separated by a short linker that can be cleaved after secretion. ANGPTL4 appears on the cell surface as the full-length form, where it can be released by heparin treatment. ANGPTL4 protein is then proteolytically cleaved by proprotein convertases (PCs), including furin, PC5/6, paired basic amino acid-cleaving enzyme 4, and PC7.
Recognizes the coiled-coil domain (CCD) of human ANGPTL4. Detects a band of ~19kDa by Western blot.
Non-hazardous
Primary Antibodies
12352203
Stable for at least 1 year after receipt when stored at -20°C.